Bipolar Electrodialyzer (EDBM)
Three chambers(triplet) Electrodialyzer BPED
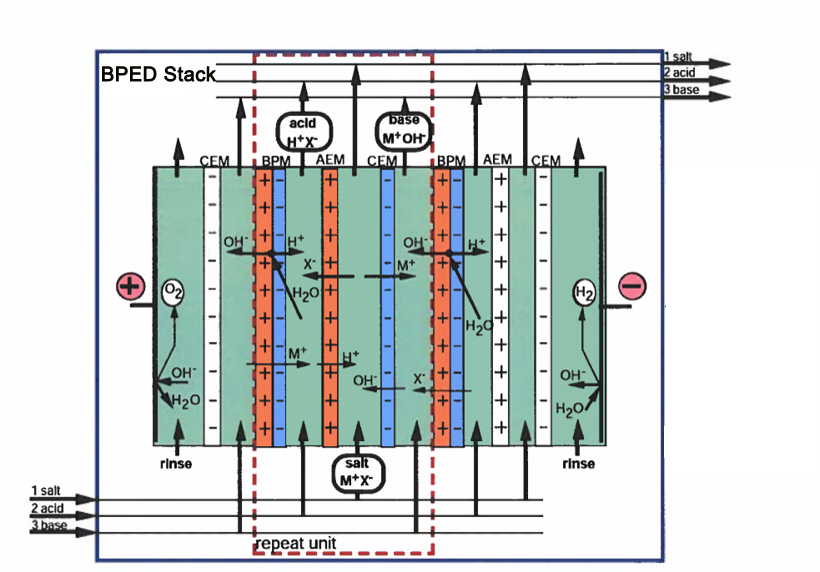
In a Bipolar Electrodialyzer (EDBM), a triplet (also called a "cell" or "repetitive unit") is the fundamental repeating unit of a 3-compartment (chambers) membrane stack. It is specifically designed to convert salts into their corresponding acids and bases through water dissociation. It consists of three specific membranes and three corresponding liquid channels that work together to convert a salt into its constituent acid and base.
Components of a Triplet
A standard three-compartment triplet consists of three different ion-exchange membranes and three flow channels:
Membranes:
Bipolar Membrane (BPM): The "engine" of the system. It consists of an anion-selective layer and a cation-selective layer together. Under an electric field, it forces water molecules to split into H+and OH- ions at its internal interface and its junction.
Anion-Exchange Membrane (AEM): Allows negatively charged ions (anions) to pass through.
Cation-Exchange Membrane (CEM): Allows positively charged ions (cations) to pass through.
Channels (Solution Compartments):
Acid Channel: Where H+ from the BPM combines with anions from the salt channel to form acid.
Alkaline/Base Channel: Where OH- from the BPM combines with cations from the salt channel to form base.
Salt/Saline Channel: Where the feed salt solution is depleted of its ions.
Working Principle
Water Dissociation: When a direct current is applied, water molecules at the interface of the BPM layers split into protons H+and hydroxide ions OH-.
Ion Migration: The applied electric field forces cations from the salt channel through the CEM into the base channel, and anions through the AEM into the acid channel.
Chemical Production: The migrated salt ions combine with the generated H+ andOH- to produce concentrated acid and base solutions.
How the Three Compartments Work
The membranes create three distinct flow channels within the triplet(3 chambers):
Salt Channel (Feed): Located between the AEM and CEM. As current flows, salt cations M+ move through the CEM toward the cathode, and salt anions X- move through the AEM toward the anode. This "desalinates" the feed stream.
Acid Channel: Located between the AEM and the cation side of the BPM. The BPM injects H+ ions into this channel, while the salt anions X- enter from the salt channel. Together, they form the acid HX.
Base Channel: Located between the CEM and the anion side of the BPM. The BPM injects OH- ions here, while salt cations M+ enter from the salt channel, forming the base MOH
Why Use a Triplet?
The triplet configuration is the "gold standard" for salt splitting for several reasons:
High Purity: Because the acid and base are formed in separate dedicated compartments, there is minimal contamination between the two.
Simultaneous Action: It performs three tasks at once: it produces acid, produces base, and dilutes the original salt stream.
Versatility: It can process inorganic salts (like Na2SO4 into h2SO4 and NaOH or organic salts (like sodium lactate into lactic acid).
Comparison: Triplet vs. Doublet
| Feature | Triplet (3-Compartment) | Doublet (2-Compartment) |
| Membranes | BPM + AEM + CEM | BPM + (AEM or CEM) |
| Output | Pure Acid and Pure Base | Acid/Salt mixture or Base/Salt mixture |
| Complexity | Higher (3 pumps/loops) | Lower (2 pumps/loops) |
| Best For | High-value chemical recovery | Simple pH adjustment |
Applications
Brine Valorization:Converting waste salt from desalination or industrial processes back into useful chemicals.
Organic Acid Production: Recovering pure organic acids (citric, acetic, lactic) from fermentation broths.
Zero Liquid Discharge (ZLD): Helping factories close the water loop by turning waste effluent back into process reagents.
Lithium Recovery: Extracting lithium from brine streams.
Organic Acid Production: Recovering high-value products like citric or formic acid.
Desalination Pre-treatment: Reducing the footprint of seawater reverse osmosis by removing boron and producing cleaning chemicals in-situ.
Practical Example 1:
A factory produces sodium sulfate (Na2SO4) waste. Using BMED, they can recycle this waste back into Sulfuric Acid (H2SO4) and Sodium Hydroxide (NaOH) to reuse in their own production line.
Practical Example 2:
in the Battery Recycling,BMED is now being used to treat the "Black Mass" from recycled batteries, recovering lithium from Li2SO4 wastewater generated during the smelting/leaching process.
Our BPED Type
BPED cells: one cell=1pcs AEM + 1pcs BPM + 1pcs CEM
50cells: generally for pilot testing
100 cells: for Industry scale
200 cells: for Industry scale
Our membrane size: 80*40cm, 110*55cm
Transportation
We will choose the suitable transportation mode and deliver the goods to you on time and safety